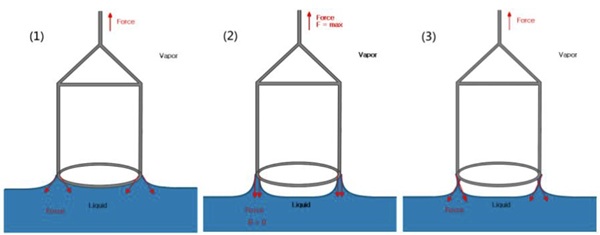
Wicking experiments are used to obtain weight pick-up information for porous solid samples. These experiments can also be used to identify unknown samples. If the sample under investigation is porous, it can be attached to the fixing device either with the solid Jig or Powder Accessory and allowed to rest at the surface of the liquid.

A wicking measurement involves the following steps:
- The sample is held at the liquid’s surface or underneath the liquid’s surface.
- The weight change as it adsorbs/absorbs liquid is monitored against time and if the density of the liquid is known the output can be displayed as volume versus time.
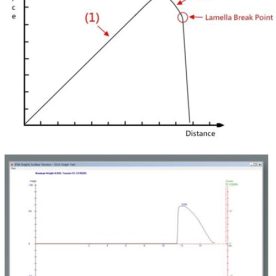
This procedure allows for comparative tests of similar porous solids against the same liquid or similar liquids against a standard porous solid to be made.
Powder Contact Angle (Washburn Method)
This test is a quantitative adsorption test by which the contact angle of an unknown liquid on a porous solid can be determined. If the liquid used in an adsorption test totally wets the porous solid (contact angle = 0), Washburn theory can be used to calculate the material constant of the porous solid. Then by using different non-wetting liquids, the quantitative contact angle can be found.

For this measurement, it is necessary to know other parameters e.g. surface tension, density, and viscosity of the liquids used. If standard test liquids are used, a further calculation can be made to obtain the porous solid’s surface energy. The method for measuring the contact angle of porous substrates relies on the test liquids penetrating the sample, sometimes called wicking behaviour.
By relating the fibres and powder jig to capillaries, the theory of Washburn who described the rising height of a liquid in a capillary, is applied:
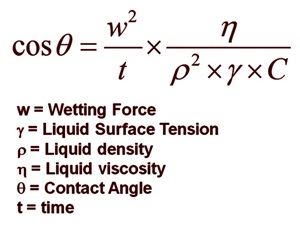
In the Washburn equation, the dimensionless constant C can be obtained by measuring with a standard liquid which totally wets the sample without reacting with it. For this purpose, n-Hexane, n-Heptane, or n-Octane can be utilised, due to their very low surface tension values. The equation also involves other physical constants for the liquid. Since they will vary when the temperature does, it is important to keep the experimental conditions as much the same as possible. This includes temperature, humidity, etc. It is also necessary to change the liquid often.
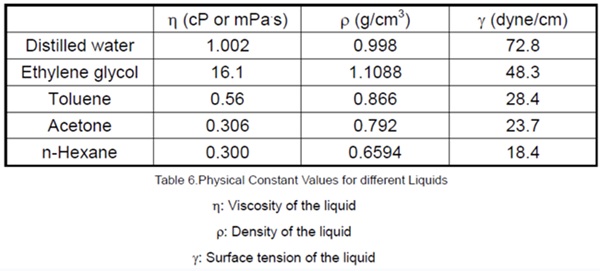
Typical physical constant values for different liquids:
Sample preparation is the most important step of the experiment since the powder is the most heterogeneous sample. The following steps should be performed in order to obtain the best representative results for the product:
- The sample that is collected from the production line or field should be representative of the batch. In general, about 0.2 % of the material should be collected for laboratory use.
- The physical properties of the samples, such as particle size, dryness and grinding, should be determined before other procedures.
- The volume of the sample should be reduced. This can be done by properly mixing the sample and dividing it into smaller portions. One commonly used technique is called Coning and Quartering.
- Measuring samples must always be prepared in equal quantities and the filling height in the powder sample holder must be at least 1 cm.
- The dimension constant, C, also represents the inner structure of the packed powder. In order to obtain repeatable C values, it is then important to keep the same packing. This requires the user to establish a packing procedure prior to the experiments by trying different packing procedures.
- It is necessary to use filter paper for experiments against both the standard and testing liquids if powder particles fall into the liquid through the bottom of the powder accessory. The filter paper should be made of high absorbent material, should be tight enough to hold the powder, and thin enough to allow fast passage of liquids.
Click here to access the DST series of analysers which offer wicking and powder contact angle measurements using the above methods.
Contact us for additional information on measurements and modalities for different surface chemistry analyses.