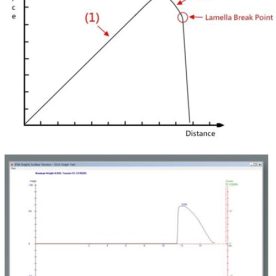
Surfactants form ordered aggregates called micelles once their concentration exceeds a threshold value. The Critical Micelle Concentration (CMC) is the point where micelles start to form. CMC measurements are used to measure the surface tension of surfactant liquids.
This can be determined from the plot of the surface tension as a function of the logarithm of surfactant concentration. As the concentration of surfactant increases there is a corresponding drop in surface tension until the CMC is reached, and at this point, the surface tension remains constant.

The test consists of running a surface tension measurement using the Du Nuoy Ring method for several different surfactant concentrations. The procedure is schematically explained below and makes use of the optional CMC kit which includes a stirrer:

The surface tension value is stored with its respective concentration and the experiment is repeated for various concentrations. The result is a master plot of surface tension vs. concentration, from which the CMC can be determined.
Conditions of influence
All the above measurements are capable of being influenced by several environmental factors and measurement conditions such as:
- Maintain Pt-Ir ring and Pt-plate: The ring and plate should always be in their original shape. When deflection occurs, the surface tension calculations are affected.
- Temperature: Surface tension is influenced by temperature. Usually, if the temperature rises, surface tension decreases.
- Measurement time:I f the probes are inserted and removed too fast, then accurate results may not be obtained.
- Stabilization time: Electronic balance is very sensitive. After turning on the power, we need to wait for more than an hour to ensure the stability of the weight. Measurements need to be performed at mechanical equilibrium.
- Density: During measurement of interfacial tension, if the density difference is not large, the tension value is small and it is difficult to get an accurate result. If the interface tension is below 10 dyne/cm, the spinning drop method is recommended.
- Surface cleaning: If the ring, plate, or any probe is contaminated or dirty, then the measurement results will not be accurate or reproducible.
- Purity of liquid: Consider the effect of contamination of liquid by ambient air and ensure the liquid purity is high and uniform across measurements.
- Surface roughness and pore size: If samples contain microscopic dust or pores on the surface, the surface tension values will change.
- Influence of adsorption: When moisture is absorbed prematurely in samples, correct measurements are difficult to obtain.
- Vibration: SEO’s DCA Series has an anti-vibration mechanism and algorithm, but too much vibration results in incorrect values measured by the force balance.
Click here to access the DCA series of analysers which offer CMC measurements using the above method.
Contact us for additional information on measurements and modalities for different surface chemistry analyses.