If you’ve ever wondered why some liquids mix perfectly while others stubbornly separate, or why a tiny drop of detergent can magically lift a grease stain, you’ve already encountered the fascinating world of surfactants.
In the high-stakes environments of research laboratories and industrial R&D across Australia and New Zealand, surfactants are the unsung heroes of formulation science. But to truly harness their power, you need to understand one specific “tipping point”: the Critical Micelle Concentration (CMC).
Whether you are developing the next breakthrough in nanotechnology instrumentation or refining a new cosmetic formula, mastering CMC determination is a game-changer. In this guide, we’ll break down what CMC is, why it matters, and the high-precision tools you need to measure it accurately.
What Exactly is Critical Micelle Concentration?
Let’s start with a quick chemistry refresher. Surfactants (surface-active agents) are unique molecules with a “split personality.” They have a hydrophilic (water-loving) head and a hydrophobic (water-fearing) tail.
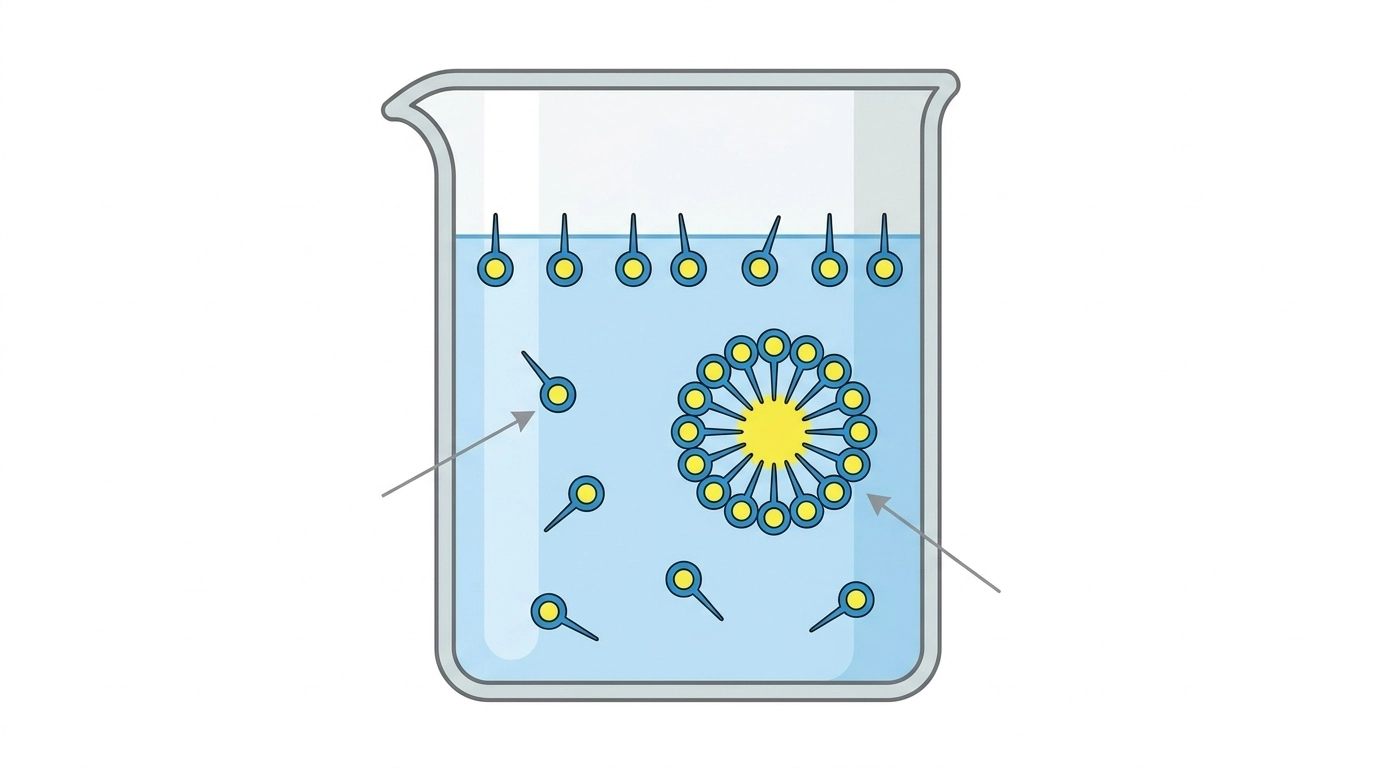
When you add a small amount of surfactant to water, these molecules rush to the surface. Their tails point toward the air (escaping the water), which reduces the surface tension of the liquid. This is why surfactants make water “wetter” and allow it to spread.
But what happens when the surface is completely crowded?
As you keep adding more surfactant, the molecules have nowhere else to go on the surface. To protect their hydrophobic tails from the water, they begin to cluster together inside the bulk of the liquid, forming spheres called micelles.
The exact concentration at which these micelles begin to form is the Critical Micelle Concentration (CMC).
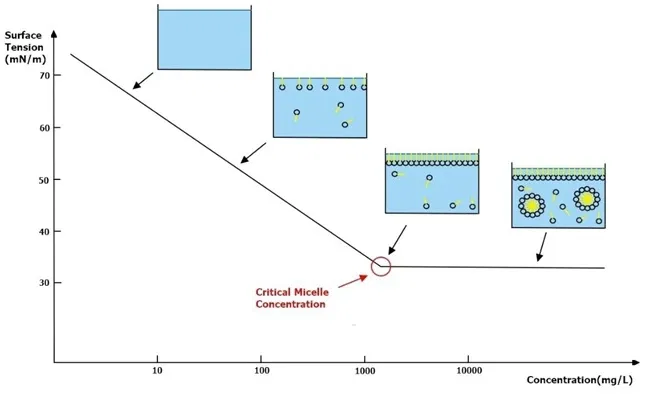
As shown in the graph above, once you reach the CMC, adding more surfactant no longer significantly reduces the surface tension. Instead, the surplus molecules just join existing micelles or form new ones.
Why CMC Matters in Your Research
In many industries, the CMC is the “magic number” that dictates how a product will perform. Here are a few ways researchers in our neck of the woods are using CMC data:
1. Cosmetic and Personal Care
From silky lotions to foaming cleansers, surfactants are everywhere in cosmetics. Knowing the CMC helps researchers create stable emulsions. If you use too little surfactant, the cream might separate; use too much, and you’re wasting expensive raw materials or risking skin irritation.
2. Pharmaceutical Research
In drug delivery, micelles can be used as “delivery vehicles.” Many drugs are hydrophobic and don’t dissolve well in the bloodstream. By encapsulating these drugs inside micelles, researchers can improve solubility and ensure the medication reaches its target effectively.
3. Food Science
Ever wondered why the oil in your salad dressing stays mixed? Emulsifiers (a type of surfactant) are the key. Food science research relies on CMC determination to ensure shelf-life stability and the perfect texture in everything from chocolate to mayonnaise.
4. Nanotechnology and Material Science
In the world of nanotechnology instrumentation, CMC is vital for synthesizing nanoparticles with specific sizes and shapes. Micelles act as “nano-reactors,” providing a controlled environment for chemical reactions at the molecular level.
How to Measure CMC with Precision
You can’t just “see” a micelle form with the naked eye. To find the CMC, you need a high-precision laboratory tensiometer.
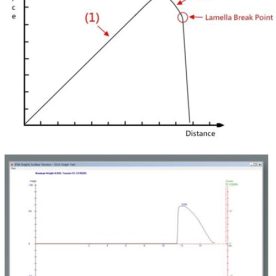
The process involves measuring the surface tension of a liquid as you progressively increase the concentration of the surfactant. By plotting these points on a graph, you’ll see a sharp “break” in the curve: that’s your CMC point.
At NanoTechnology Solutions, we recommend the DST and DCA series for these tasks. These instruments are designed to handle the delicate nuances of surface tension measurements with automated accuracy.

The DST-30, for example, features an automated measurement protocol that removes human error from the equation. It even includes an anti-vibration sample chamber, which is essential when you’re measuring at the milli-Newton per meter (mN/m) level.
Choosing Your Method: Ring vs. Plate
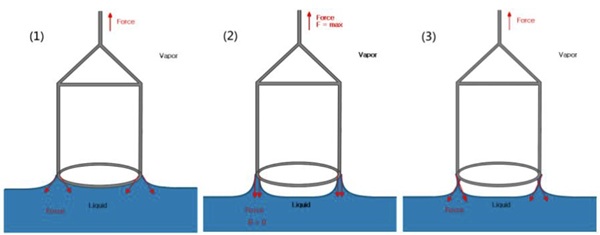
When it comes to interfacial tension measurement and CMC determination, you generally have two main choices for your probe: the Du Noüy Ring or the Wilhelmy Plate.
The Du Noüy Ring Method
This is the “classic” method and remains a staple in many Australian labs. It involves a platinum-iridium ring that is lowered into the liquid and then slowly pulled through the surface. The instrument measures the maximum force required to “stretch” the liquid film before the ring breaks free.

Pros:
- Well-established and standardized (great for historical data).
- Excellent for measuring interfacial tension between two liquids.
Cons:
- Requires mathematical “correction factors” because the weight of the liquid pulled up by the ring isn’t perfectly cylindrical.
- The ring is delicate and can be easily bent if not handled with care.
The Wilhelmy Plate Method
In this method, a thin rectangular plate (often made of platinum or even specialized paper) is lowered into the liquid until it just touches the surface. The instrument measures the force exerted on the plate due to the “wetting” of the surface.
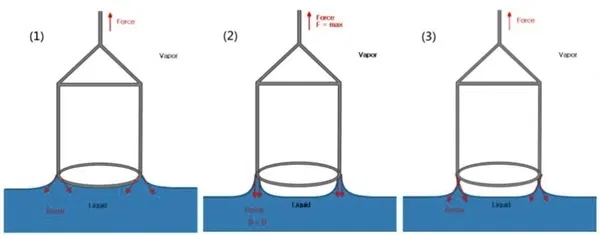
Pros:
- No correction factors are needed (it’s a direct measurement).
- Ideal for monitoring surface tension over a long period.
- Less disruptive to the liquid surface, which is crucial when measuring surfactants that take time to reach equilibrium.
Cons:
- Requires the plate to be perfectly clean and the contact angle to be exactly zero (complete wetting).
Pro-Tip: Don’t Forget Density!
A common mistake in CMC determination is ignoring the density of the liquid. Many surface tension calculations require an accurate density value to provide a precise result. High-end tensiometers, like our DCA series, can often perform density measurements using a specialized probe, ensuring your CMC data is as accurate as possible.
Why Technical Support is Your Secret Weapon
Investing in a laboratory tensiometer is a big step for any research facility. But the hardware is only half the battle.
In Australia and New Zealand, researchers often face unique challenges, from sourcing specialized calibration fluids to finding local technical support when a question arises mid-experiment. At NanoTechnology Solutions, we don’t just ship you a box; we provide a proven framework for selecting the right analytical tools and offer the technical backup you need to keep your lab running smoothly.
Wrapping Up: Mastering Your Surfactant Analysis
Understanding Critical Micelle Concentration is more than just a box-ticking exercise in a lab report. It’s the key to unlocking better product performance, more efficient drug delivery, and stable material formulations.
By choosing the right method: whether it’s the robust Du Noüy ring or the precise Wilhelmy plate: and utilizing high-precision tools like the DST-30, you can turn a complex chemical transition into a repeatable, reliable data point.
Ready to upgrade your surface analysis? Whether you’re in Sydney, Auckland, or anywhere in between, our team at NanoTechnology Solutions is here to help you find the perfect tensiometer for your specific research needs. Contact us today to discuss how we can support your next project!