Researchers at leading universities and industrial laboratories worldwide face a common challenge in sample preparation: selecting the wrong process gas in the plasma cleaning process can compromise surface characterisation results, damage delicate substrates, or leave residual contamination that undermines months of experimental work. The decision between oxygen, hydrogen, and argon plasma determines whether a surface preparation succeeds or fails.
Why Plasma Cleaning Gas Selection Matters
Plasma cleaning has become indispensable in nanotechnology research and precision manufacturing. Unlike wet chemical methods, plasma cleaning processes clean surfaces at the molecular level without introducing liquid contaminants or requiring extensive drying protocols. The technology creates an ionised gas state that interacts with surfaces through chemical reactions or physical bombardment, depending on the gas selected.
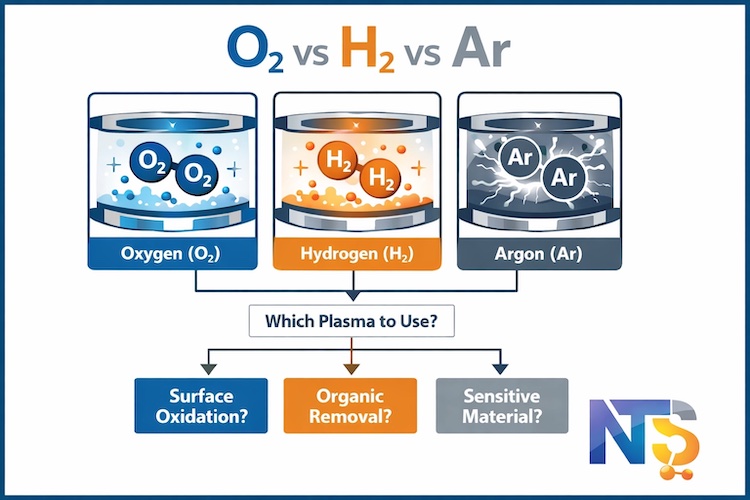
The three primary plasma gases: O₂, H₂, and Ar: each operate through fundamentally different mechanisms. Selecting the appropriate gas requires understanding not just what needs removal, but also the substrate composition, desired surface properties, and downstream processing requirements.
Oxygen Plasma: The Organic Contaminant Specialist
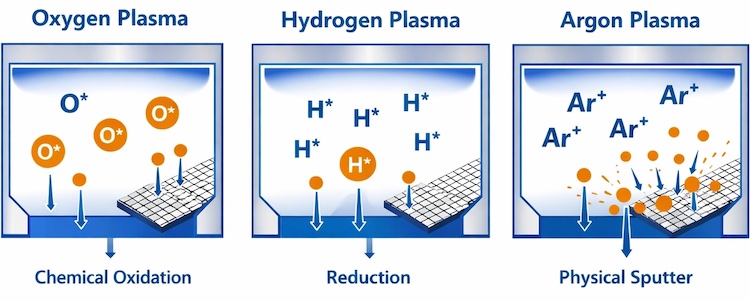
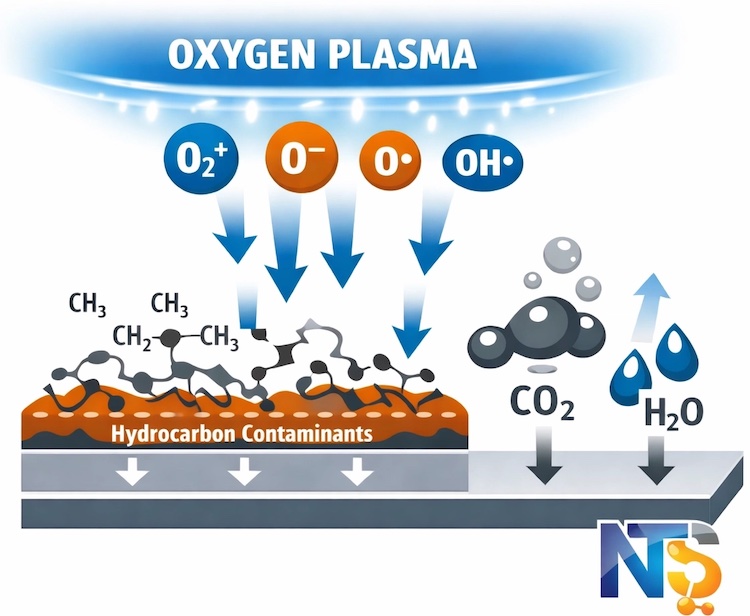
Oxygen plasma functions as a highly oxidative environment that excels at eliminating organic contamination. The process breaks down hydrocarbons, oils, photoresist residues, and carbon-based materials through chemical reactions analogous to controlled combustion at the molecular level.
The reactive oxygen species generated in the plasma: including atomic oxygen, ozone, and oxygen ions: attack organic molecules by breaking carbon-hydrogen and carbon-carbon bonds. This makes oxygen plasma the fastest and most effective option for removing oils, fingerprints, and other organic contaminants commonly encountered in laboratory environments.
Beyond cleaning, O₂ plasma introduces oxygen-containing functional groups to surfaces. Hydroxyl (-OH) and carboxyl (-COOH) groups form on treated surfaces, dramatically increasing surface energy and improving wettability. This surface activation proves critical for applications requiring strong adhesion: wire bonding, thin film deposition, wafer bonding, and optical coating processes all benefit from oxygen plasma pre-treatment.
Material compatibility represents the primary constraint. Oxygen plasma aggressively oxidises metals like copper, silver, and aluminium. Researchers working with these materials must avoid oxygen plasma or carefully control exposure times to prevent unwanted oxide formation that can alter electrical properties or introduce measurement artefacts in surface characterisation.
Hydrogen Plasma: The Reduction Chemistry Solution
Hydrogen plasma operates in the opposite chemical direction from oxygen, creating a reductive environment rather than an oxidative one. This fundamental difference makes H₂ plasma the preferred choice for removing oxide layers from metal surfaces and restoring them to their pure, elemental state.
The mechanism involves reactive hydrogen species that chemically reduce metal oxides. For example, copper oxide (CuO) exposed to hydrogen plasma undergoes reduction back to pure copper metal, with water vapour as the byproduct. This oxide removal capability proves essential in semiconductor processing, metal surface preparation, and applications where oxide layers interfere with electrical contact or catalytic activity.
Hydrogen plasma can also remove organic contaminants, though through different chemistry than oxygen. The reducing environment makes it suitable for materials that cannot tolerate oxidative conditions. Additionally, H₂ plasma processes typically operate at lower temperatures than many alternative cleaning methods, protecting heat-sensitive substrates like polymers and biological samples.
Safety protocols require careful attention. Hydrogen gas presents flammability concerns that demand proper ventilation, gas handling procedures, and system design considerations not required for inert gases.
Argon Plasma: The Physical Cleaning Method
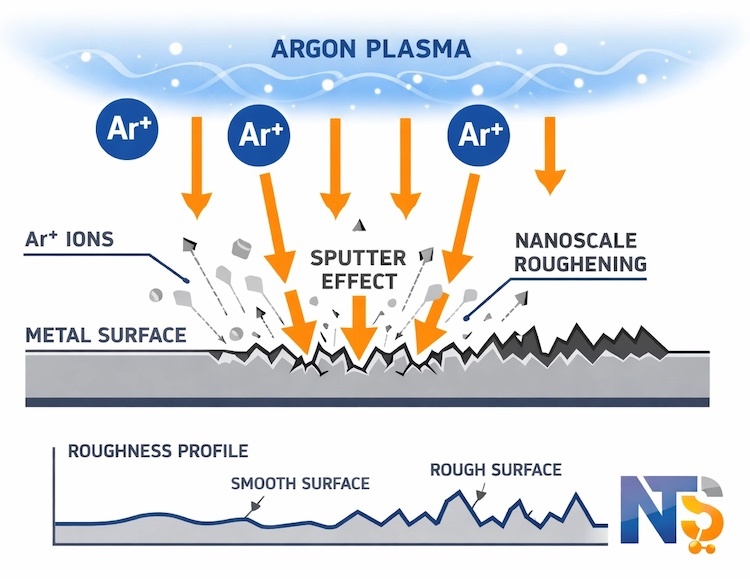
Argon plasma takes a fundamentally different approach to surface cleaning. As an inert gas, argon does not chemically react with surfaces or contaminants. Instead, the cleaning mechanism relies entirely on physical bombardment.
Heavy argon ions accelerated in the plasma field strike surfaces with significant kinetic energy. This creates a micro-sandblasting effect at the atomic scale, physically dislodging loosely bound contaminants through momentum transfer. The process simultaneously roughens surfaces at the nanometer scale, increasing surface area and creating mechanical anchoring sites that enhance adhesion for subsequent coating or bonding steps.
The inert nature of argon plasma makes it the safest choice for chemically sensitive materials. Samples that would oxidise in oxygen plasma or react unpredictably with hydrogen plasma can be cleaned with Ar plasma without risk of chemical modification. This includes exotic materials, multilayer structures, and samples with mixed compositions where reactive gas chemistry might produce unwanted side reactions.
Processing speed represents the primary limitation. Argon plasma cleaning proceeds more slowly than reactive gas alternatives. The purely physical mechanism cannot efficiently break down strongly bonded organic contaminants like polymerised residues. For these materials, researchers often employ sequential processing: oxygen plasma for organic removal followed by argon plasma for surface roughening and final conditioning.
The Gas Selection Framework
The optimal plasma cleaning gas depends on multiple factors that researchers must evaluate systematically:
For organic contamination removal: Oxygen plasma provides the fastest and most complete cleaning of oils, hydrocarbons, and carbon residues. The chemical breakdown of organic molecules proceeds rapidly, making O₂ plasma the default choice when material compatibility permits.
For oxide layer removal: Hydrogen plasma uniquely reduces metal oxides back to pure metal surfaces. This capability has no substitute when oxide layers must be eliminated rather than simply cleaned.
For chemically sensitive materials: Argon plasma offers cleaning without chemical modification. Materials that would oxidise, reduce, or otherwise react with oxygen or hydrogen require the inert approach.
For surface activation and adhesion improvement: Both oxygen plasma and argon plasma enhance surface energy, but through different mechanisms. O₂ plasma chemically functionalises surfaces, while Ar plasma physically roughens them. The choice depends on whether the material tolerates oxidation.
For multi-contaminant scenarios: Sequential processing or gas mixtures address complex cleaning needs. An oxygen plasma step removes organics, followed by argon plasma for final conditioning and roughening. Alternatively, oxygen-argon mixtures combine chemical and physical cleaning in a single process.
Operational Considerations Beyond Gas Selection
System pressure influences cleaning mechanisms and safety. Vacuum systems provide precise control over plasma parameters but require vacuum infrastructure. Atmospheric pressure plasma systems eliminate vacuum requirements and reduce electrostatic discharge risks for sensitive electronics, though they limit process flexibility.
Power settings and exposure time must be optimised for each application. Insufficient treatment leaves contamination, while excessive exposure can damage surfaces through over-etching or unwanted chemical modification.
Gas purity affects results significantly. Trace contaminants in process gases can introduce unexpected surface chemistry or leave residues that compromise subsequent processing steps.
Advanced Plasma Cleaning Systems for Precision Research
NanoTechnology Solutions provides plasma cleaning systems that address the full spectrum of surface preparation requirements in modern nanotechnology research. The Tergeo series plasma cleaners support oxygen, hydrogen, and argon plasma processes with precise control over power, pressure, and treatment time.
In a practical workflow, plasma cleaning sits alongside other controlled variables in the lab setup: stable benches, appropriate handling procedures, and effective vibration isolation (including optical tables) when instrumentation is sensitive to nanometre-scale motion.
These systems enable researchers to implement the gas selection framework systematically: testing different plasma chemistries, optimising parameters for specific materials, and achieving reproducible surface preparation results. The ability to switch between gases within a single platform allows laboratories to maintain multiple plasma cleaning protocols without dedicating separate equipment to each chemistry.
Advanced features include programmable recipes for consistent batch-to-batch processing, gentle plasma modes for delicate samples, and compatibility with sample geometries from small coupons to large substrates. Integration capabilities allow plasma cleaning steps to fit seamlessly into complex sample preparation workflows involving multiple surface characterisation techniques, preparing samples for high-resolution characterisation like SEM or an AFM (atomic force microscope) where surface cleanliness and surface energy directly influence measurement stability.
Making the Right Choice
The decision between oxygen, hydrogen, and argon plasma ultimately rests on understanding the specific requirements of each application. Organic contamination on oxidation-resistant materials points toward oxygen plasma. Metal oxide removal demands hydrogen plasma. Chemically sensitive samples require argon plasma‘s inert approach.
Many research workflows benefit from maintaining access to all three options. Surface preparation often involves sequential steps: oxygen plasma for initial cleaning, followed by argon plasma for surface conditioning, or hydrogen plasma for oxide removal followed by controlled re-oxidation in oxygen plasma to form precise oxide layers.
The plasma cleaning gas selection framework provides a systematic approach to surface preparation that eliminates guesswork and optimises results. By matching plasma chemistry to material properties and cleaning objectives, researchers achieve the pristine surfaces that precision nanotechnology applications demand.